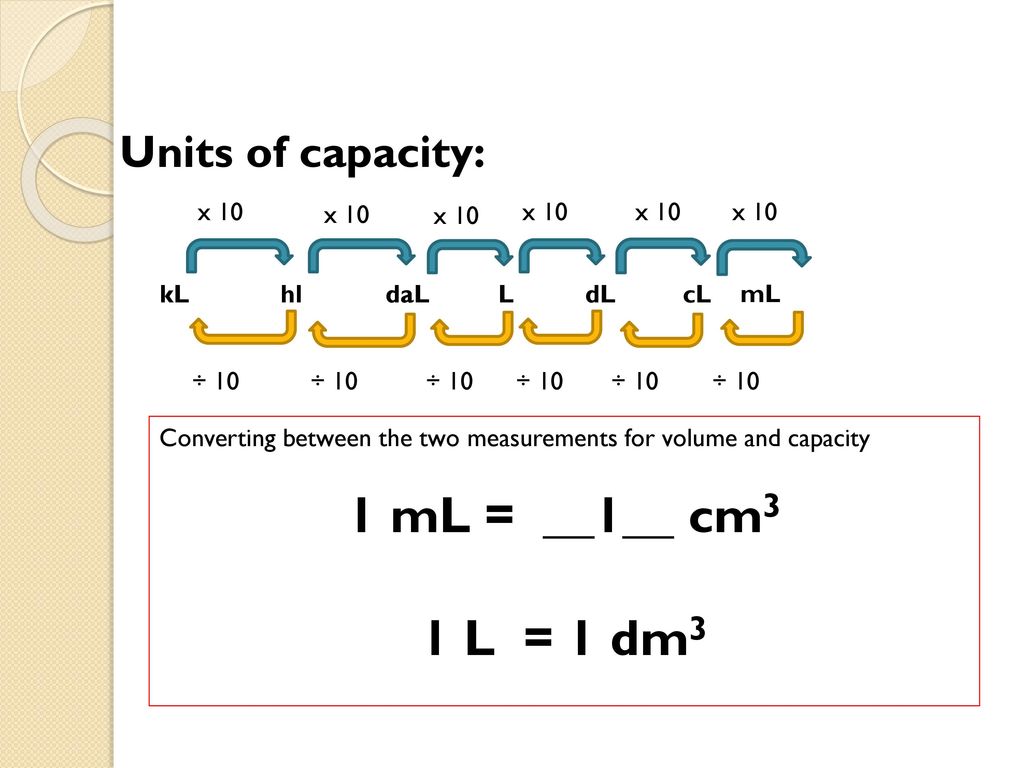
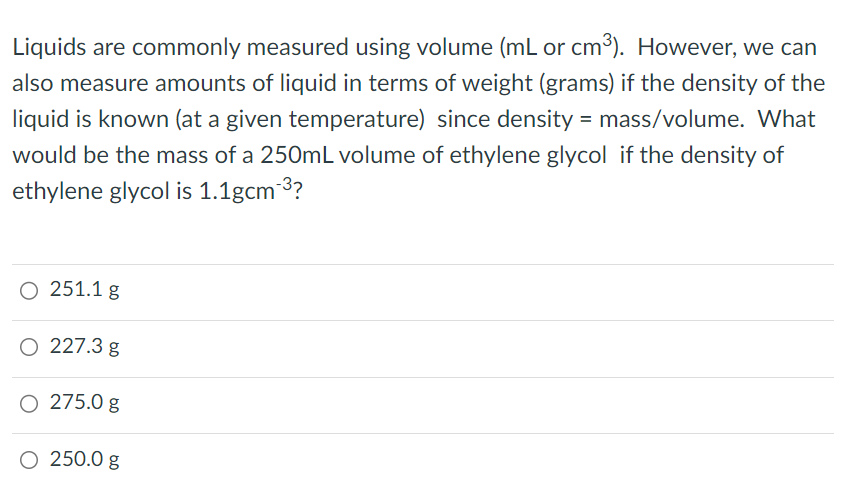1.07 Units of volume and capacity | Year 12 Maths | Australian Curriculum 12 Essential Mathematics - 2020 Edition | Mathspace
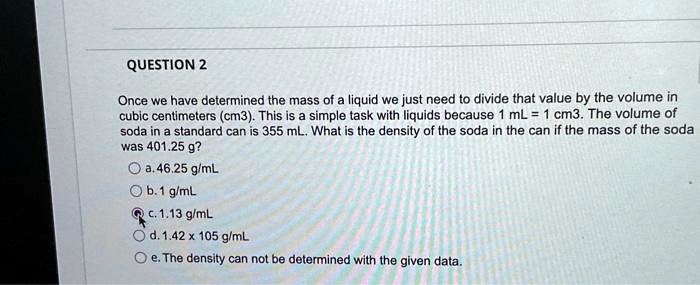
SOLVED: Once we have determined the mass of a liquid, we just need to divide that value by the volume in cubic centimeters (cm^3). This is a simple task with liquids because
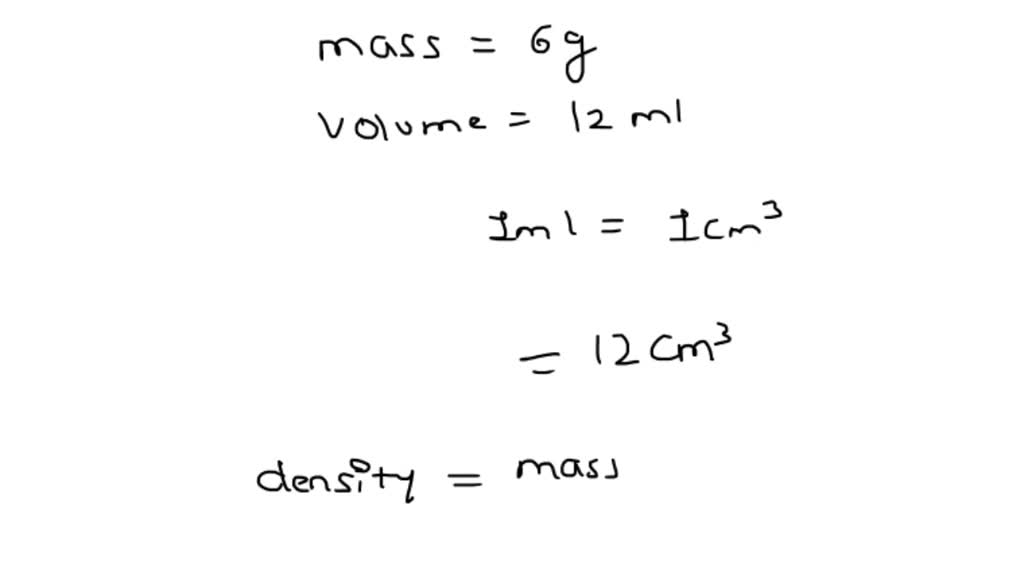
SOLVED: A sample of an unknown liquid has a volume of 12.0 mL and a mass of 6 g. What is its density? A. 2 g/cm3 B. 0.5 g/cm C. 5.0 g/cm3

SOLVED: Suppose you have 50.0 cm³ of a substance with a density of 30.0 g/L and you want to determine the mass of the substance. The calculation can be broken down into