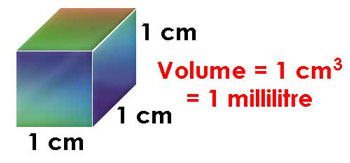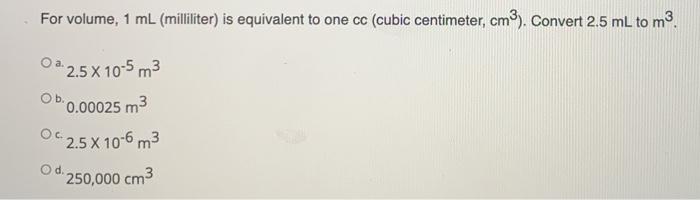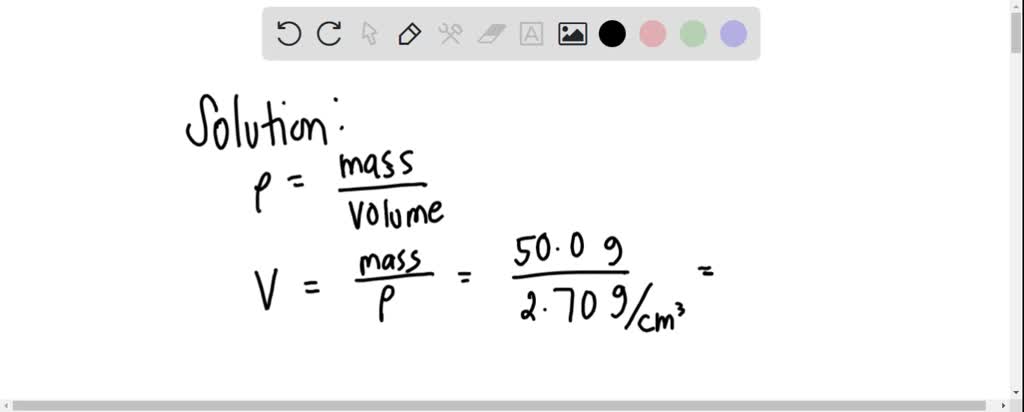
SOLVED: Density can be used as a conversion factor to convert between mass (g) and volume (mL or cm3 ). Knowing that the density of aluminum is 2.70 g/ cm3 , calculate the

1.07 Units of volume and capacity | Year 12 Maths | Australian Curriculum 12 Essential Mathematics - 2020 Edition | Mathspace

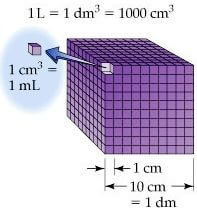
SOLVED: Units of volume (i.e., m³, cm³) can be converted to units of capacity (i.e., mL, L) using the bridge: 1 dm³ = 1 L. Volume Capacity Mm³ km³ hm³ dam³ m³




-to-Grams-(g)-Step-12-Version-4.jpg)