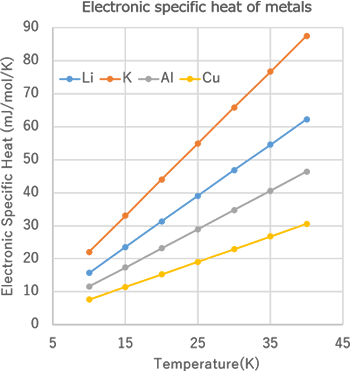
Electronic specific heat analysis of metals - J-OCTA Case Studies | CAE Solutions - JSOL Corporation
![PDF] A NEW CORRELATION FOR THE SPECIFIC HEAT OF METALS, METAL OXIDES AND METAL FLUORIDES AS A FUNCTION OF TEMPERATURE | Semantic Scholar PDF] A NEW CORRELATION FOR THE SPECIFIC HEAT OF METALS, METAL OXIDES AND METAL FLUORIDES AS A FUNCTION OF TEMPERATURE | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b0d76592f078aeeb6e66f500b45a1f101c6fe150/6-Table3-1.png)
PDF] A NEW CORRELATION FOR THE SPECIFIC HEAT OF METALS, METAL OXIDES AND METAL FLUORIDES AS A FUNCTION OF TEMPERATURE | Semantic Scholar
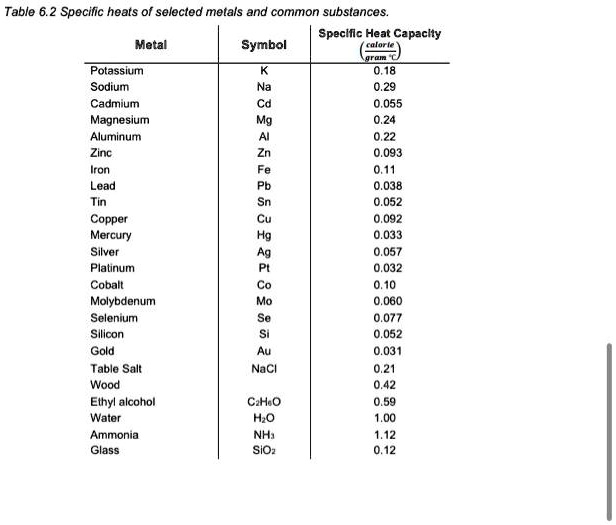
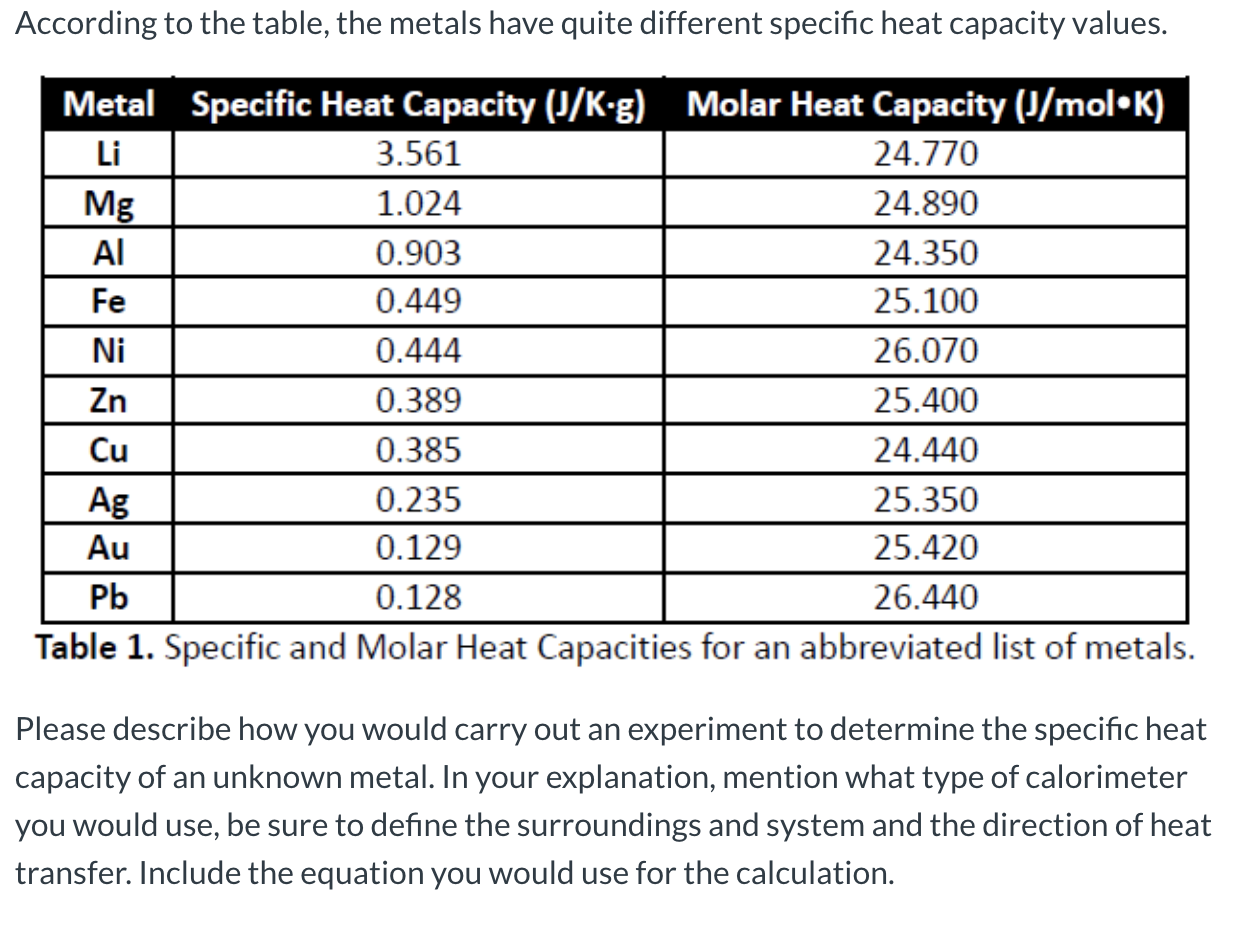
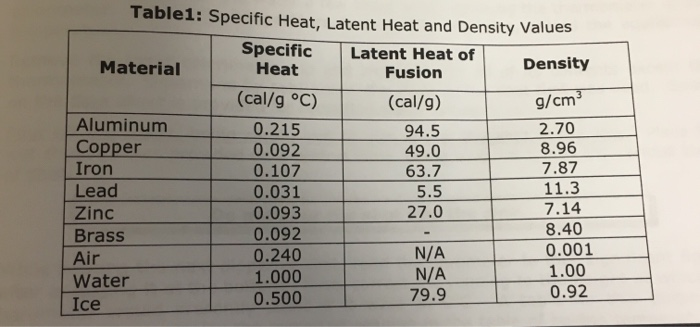
SOLVED: Table 6.2: Specific heats of selected metals and common substances Metal Symbol Specific Heat Capacity Potassium K 0.18 Sodium Na 0.55 Cadmium Cd 0.93 Magnesium Mg 0.11 Aluminum Al 0.038 Zinc
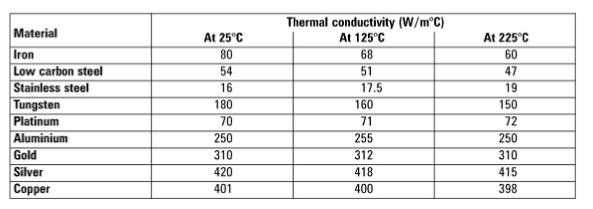
Which metal achieves the highest temperature when adding energy? Aluminum, Copper, or Silver? | CIDER

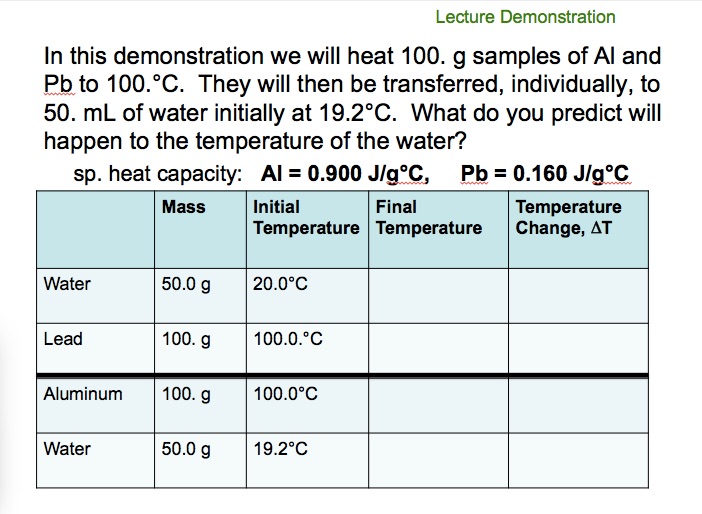
To measure the specific heat capacity of a metal (solid block method) Physics Homework Help, Physics Assignments and Projects Help, Assignments Tutors online

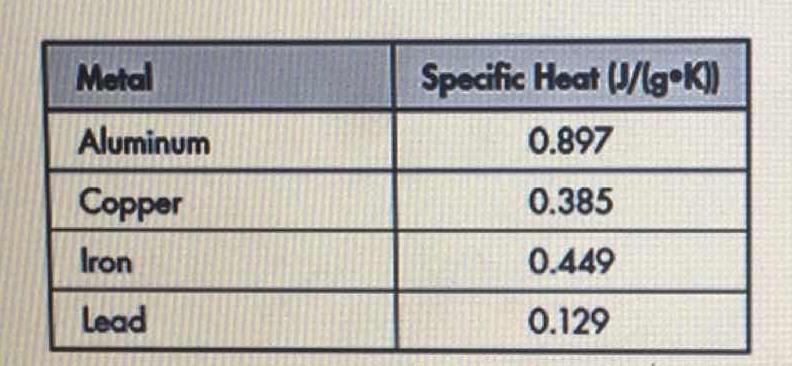
LAB: Specific Heat of a Metal. Prelab question: MetalSpecific Heat (J/g ºC) Aluminum0.91 Iron0.46 Lead0.13 Silver0.23 Tin0.21 Titanium0.54 Zinc0.39 A. - ppt download
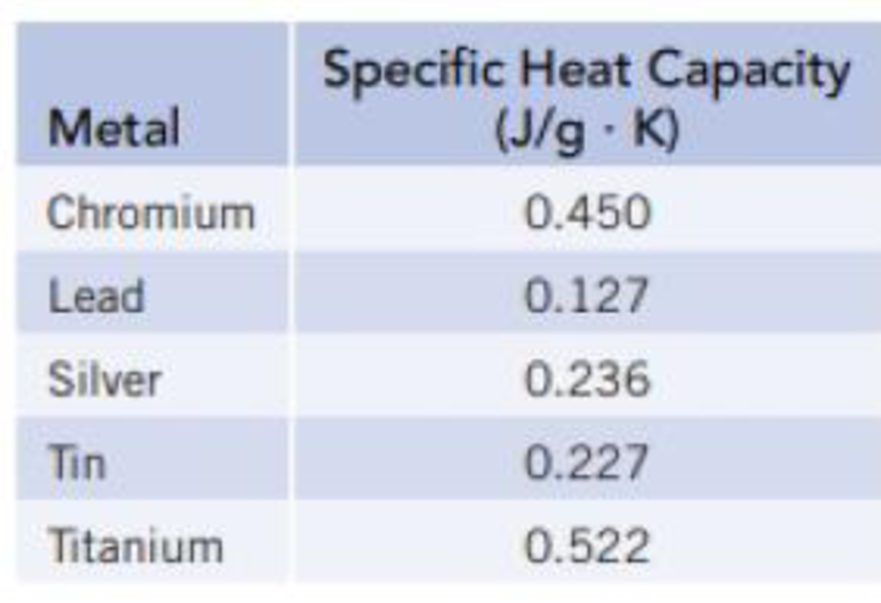
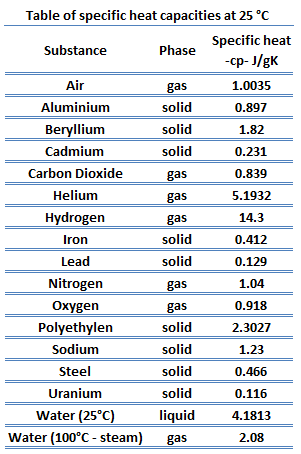
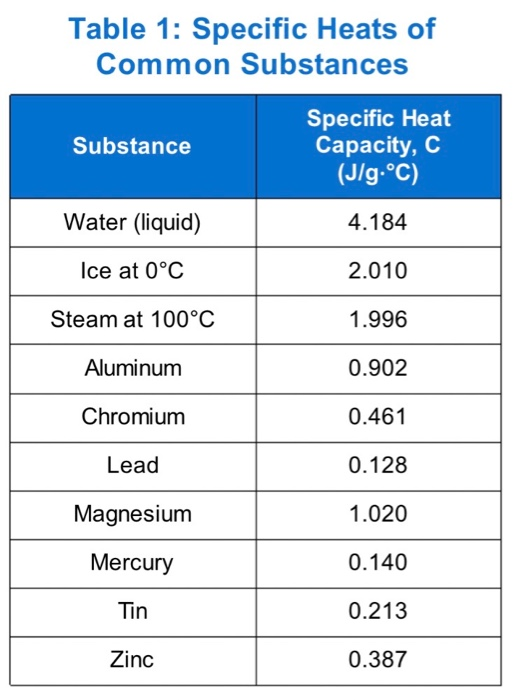
Prepare a graph of specific heat capacities for metals versus their atomic weights. Combine the data in Figure 5.4 and the values in the following table. What is the relationship between specific

Specific heat capacity of metals at low temperatures.(1 cal = 4.18 J)... | Download Scientific Diagram