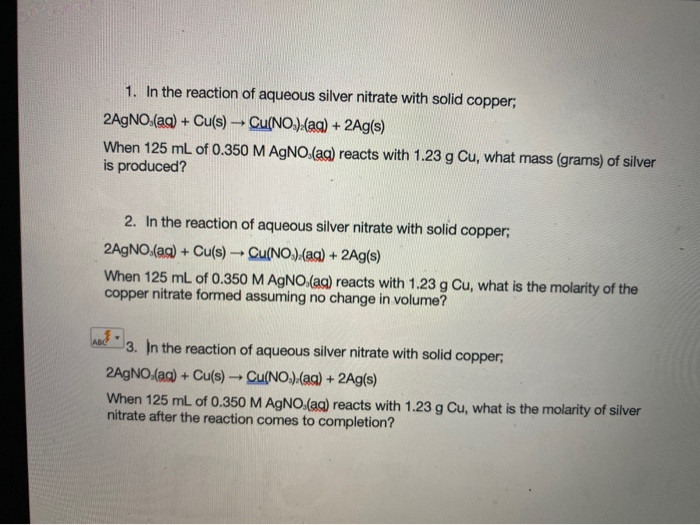
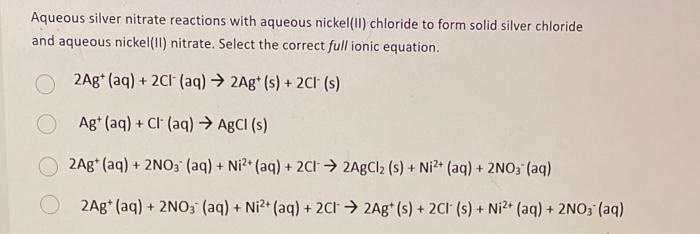SOLVED: When aqueous solutions of silver nitrate and calcium chloride are mixed, silver chloride precipitates as an insoluble solid while calcium nitrate remains in solution. In one experiment, 18.0 ml of aqueous

Flasks containing 1mM silver nitrate aqueous solution without (A) and... | Download Scientific Diagram

Silver nitrate solution is gradually added to an aqueous solution containing 0.01 M each of chloride

The video shows the addition of aqueous sodium chloride to a solution of aqueous silver nitrate. Write the - brainly.com
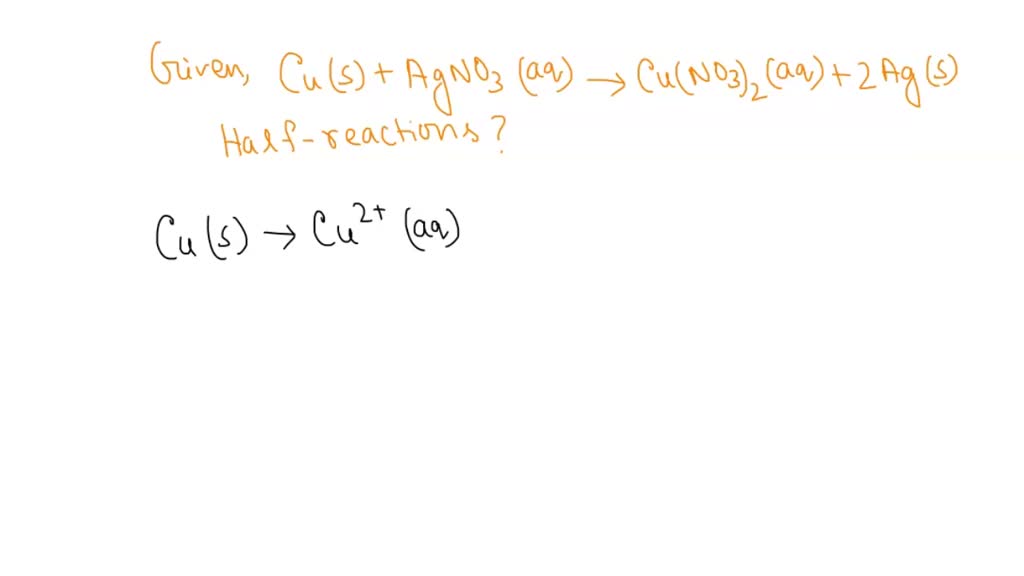
SOLVED: The reaction between solid copper and aqueous silver nitrate produces solid silver and a blue solution, as represented by the balanced equation shown below: Cu(s) + 2AgNO3(aq) -> Cu(NO3)2(aq) + 2Ag(s)

Amazon.com: Silver Nitrate - Aqueous | Part 2 of 2 for Making Silver Thiosulfate to Breed Feminized Seeds : Industrial & Scientific

science chemistry precipitation reaction silver chloride sodium chloride | Fundamental Photographs - The Art of Science

SOLVED: The reaction between solid copper and aqueous silver nitrate produces solid silver and a blue solution, as represented by the balanced equation shown below: Cu(s) + 2AgNO3(aq) -> Cu(NO3)2(aq) + 2Ag(s)

Sample tubes containing silver nitrate solution (1 mM) (A) Aqueous leaf... | Download Scientific Diagram




![Telugu] Aqueous silver nitrate is subjected to electrolysis, using pl Telugu] Aqueous silver nitrate is subjected to electrolysis, using pl](https://static.doubtnut.com/ss/web/4370988.webp)